9. Quantum Mechanics
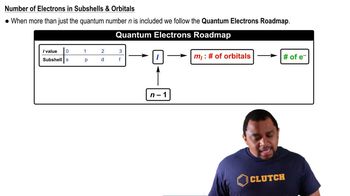
Quantum Numbers: Number of Electrons
Problem 72
Textbook Question
Textbook QuestionWhat is the maximum number of electrons in an atom that can have the following quantum numbers? (a) n = 3, ml = -1; (b) n = 4, l = 2; (c) n = 4, l = 3, ml = -2; (d) n = 5, l = 2, ml = 0.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
2585
views
1
comments
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos