23. Chemistry of the Nonmetals
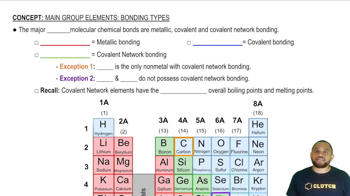
Main Group Elements: Bonding Types
Problem 22.29a
Textbook Question
Textbook QuestionConsider the six second- and third-row elements in groups 4A–6A of the periodic table:
<IMAGE>
Possible structures for the binary fluorides of each of these elements in its highest oxidation state are shown below.
<IMAGE>
(a) Identify the nonfluorine atom in each case, and write the molecular formula of each fluoride.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
0m:0sPlay a video:
19
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos