20. Electrochemistry
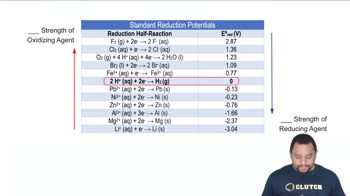
Standard Reduction Potentials
Problem 43b
Textbook Question
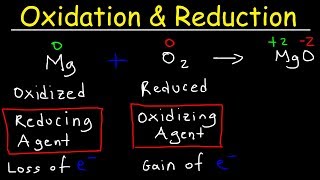
Textbook QuestionFrom each of the following pairs of substances, use data in Appendix E to choose the one that is the stronger reducing agent: (a) Fe(s) or Mg(s)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
336
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos