Determine whether or not each metal dissolves in 1 M HNO3. For those metals that do dissolve, write a balanced redox reaction showing what happens when the metal dissolves. a. Cu b. Au

Which metal can be oxidized with an Sn2+ solution but not with an Fe2+ solution?
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Oxidation and Reduction
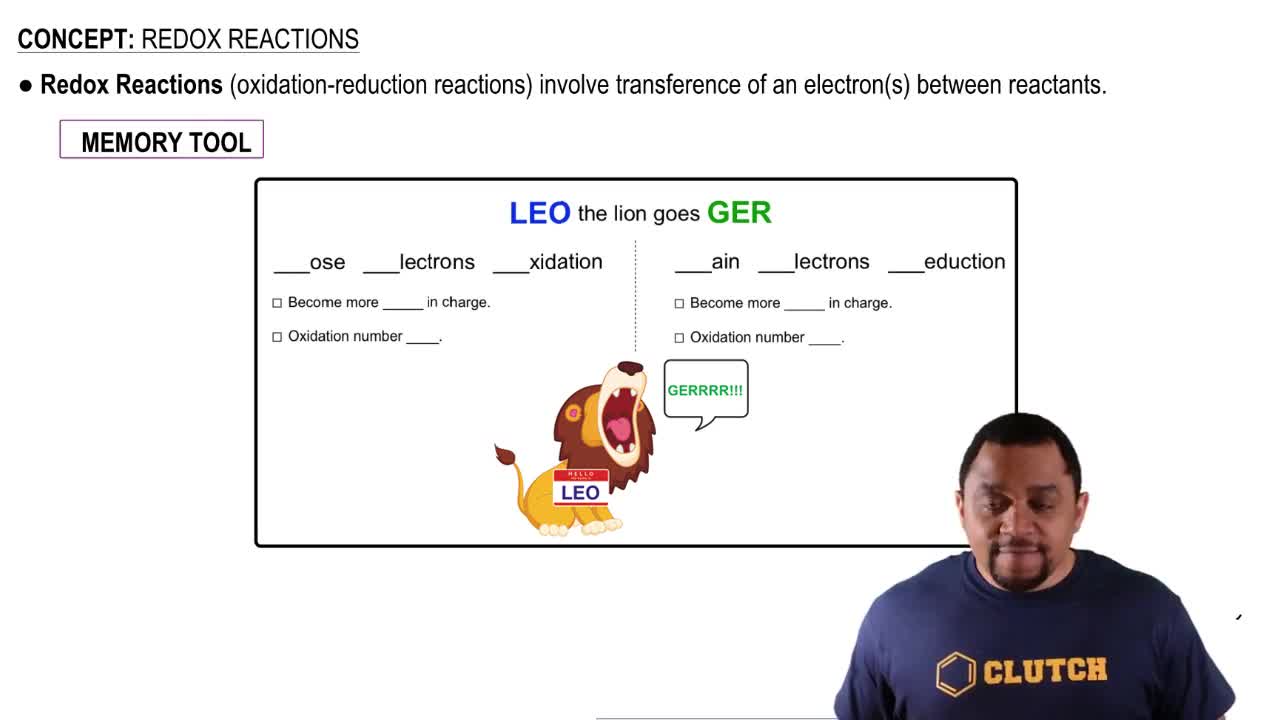
Electrochemical Series

Metal Reactivity

Determine whether or not each metal dissolves in 1 M HCl. For those metals that do dissolve, write a balanced redox reaction showing what happens when the metal dissolves. a. Al b. Ag c. Pb
Which metal could you use to reduce Mn2+ ions but not Mg2+ ions?
Determine whether or not each redox reaction occurs spontaneously in the forward direction.
a. Ca2+(aq) + Zn(s) → Ca(s) + Zn2+(aq)
b. 2 Ag+(aq) + Ni(s) → 2 Ag(s) + Ni2+(aq)
c. Fe(s) + Mn2+(aq) → Fe2+(aq) + Mn(s)
d. 2 Al(s) + 3 Pb2+(aq) → 2 Al3+(aq) + 3 Pb(s)
Determine whether or not each metal dissolves in 1 M HCl. For those metals that do dissolve, write a balanced redox reaction showing what happens when the metal dissolves. a. Cu b. Fe c. Au
Determine whether or not each redox reaction occurs spontaneously in the forward direction.
a. Ni(s) + Zn2+(aq) → Ni2+(aq) + Zn(s)
b. Ni(s) + Pb2+(aq) → Ni2+(aq) + Pb(s)
c. Al(s) + 3 Ag+(aq) → Al3+(aq) + 3 Ag(s)
d. Pb(s) + Mn2+(aq) → Pb2+(aq) + Mn(s)
