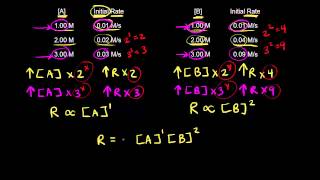15. Chemical Kinetics
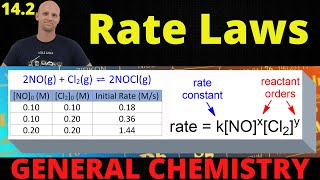
Rate Law
Multiple Choice
Multiple ChoiceFor the reaction 2 NOCl (g) → 2 NO (g) + Cl2 (g),
the experimental rate law is: rate = k[NOCl]2[NO]-1
Determine if the following mechanism is valid.
Step 1: NOCl (g) ⇌ NO (g) + Cl (g) (fast)
Step 2: NOCl (g) + Cl (g) → NO (g) + Cl2 (g) (slow)
A
No, because it has doesn't contain [Cl]
B
No, because it is not based on the slow step
C
Yes, because it contains both reactants of the slow step
D
Yes, because it reflects the stoichiometry of the overall reaction
E
Yes, because the reversible reaction was used to eliminate the intermediate
from the rate law
329
views
Related Videos
Related Practice
Showing 1 of 12 videos