3. Chemical Reactions
Limiting Reagent
Problem 74
Textbook Question
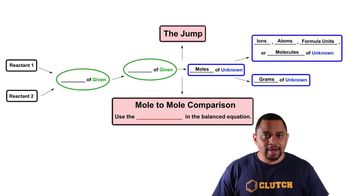
Textbook QuestionAssume that you have 1.39 mol of H2 and 3.44 mol of N2. How many grams of ammonia (NH3) can you make, and how many grams of which reactant will be left over? 3 H2 + N2 --> NH3
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
1259
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos