15. Chemical Kinetics
Half-Life
Problem 41c
Textbook Question
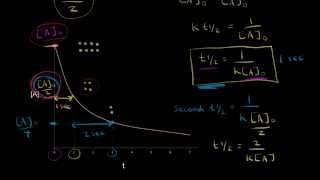
Textbook Question(a) The gas-phase decomposition of SO2Cl2, SO2Cl21g2 ¡SO21g2 + Cl21g2, is first order in SO2Cl2. At 600 K the half-life for this process is 2.3 * 105 s. What is the rate constant at this temperature?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
1413
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos