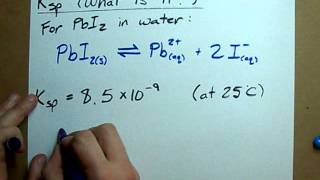18. Aqueous Equilibrium
Solubility Product Constant: Ksp
Problem 53
Textbook Question
Textbook Question(a) I f t he molar solubility of CaF2 at 35 C i s 1.24 * 10-3 mol>L, what is Ksp at this temperature?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
965
views
1
rank
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos