13. Liquids, Solids & Intermolecular Forces
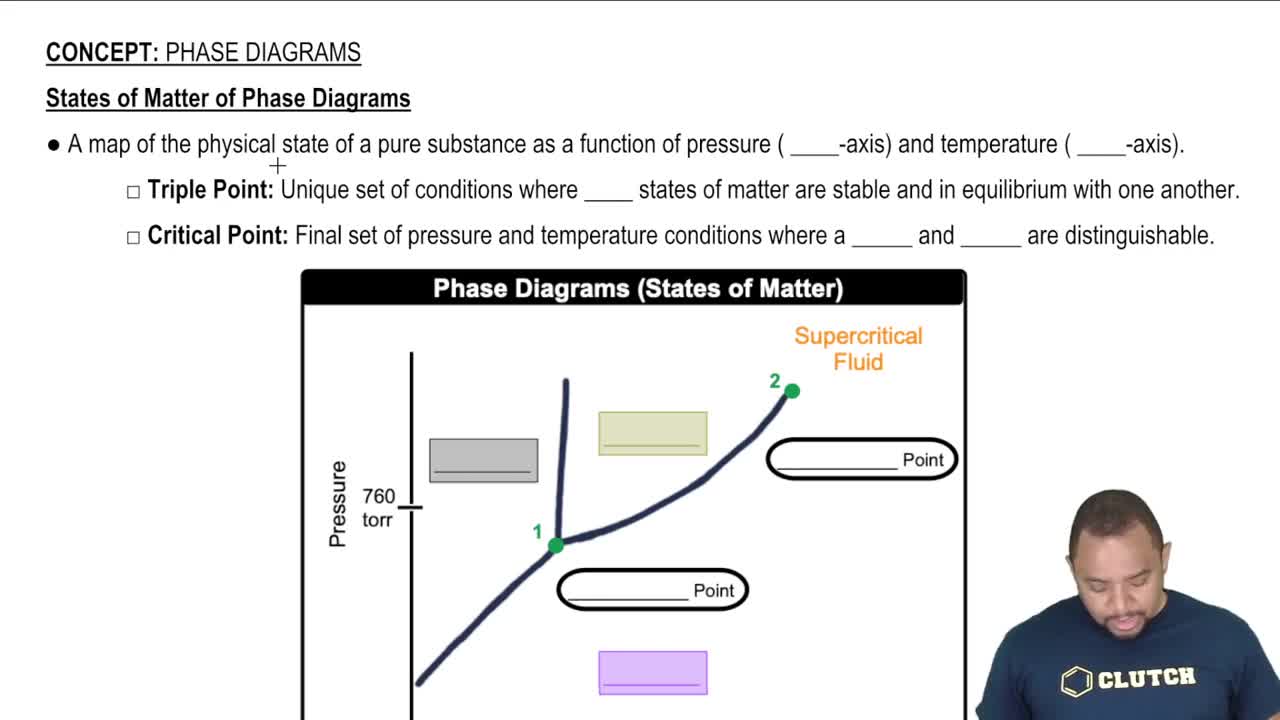
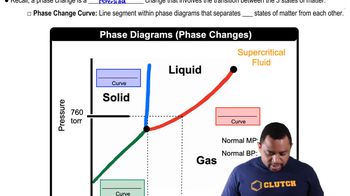
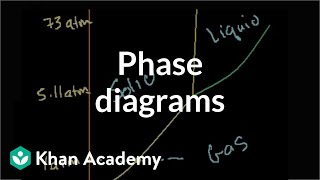
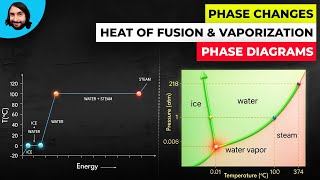
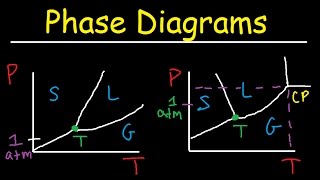
Phase Diagrams
Problem 94
Textbook Question
Textbook QuestionLiquid butane (C4H10) is stored in cylinders to be used as a fuel. The normal boiling point of butane is listed as -0.5 °C. (b) Suppose the valve to the tank is opened and a few liters of butane are allowed to escape rapidly. What do you expect would happen to the temperature of the remaining liquid butane in the tank? Explain.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
1746
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos