A voltaic cell that uses the reaction PdCl42-(aq) + Cd(s) → Pd(s) + 4 Cl-(aq) + Cd2+(aq) has a measured standard cell potential of +1.03 V. (c) Sketch the voltaic cell, label the anode and cathode, and indicate the direction of electron flow

Using standard reduction potentials (Appendix E), calculate the standard emf for each of the following reactions: (c) Fe1s2 + 2 Fe3+1aq2 ¡ 3 Fe2+1aq2
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Standard Reduction Potentials

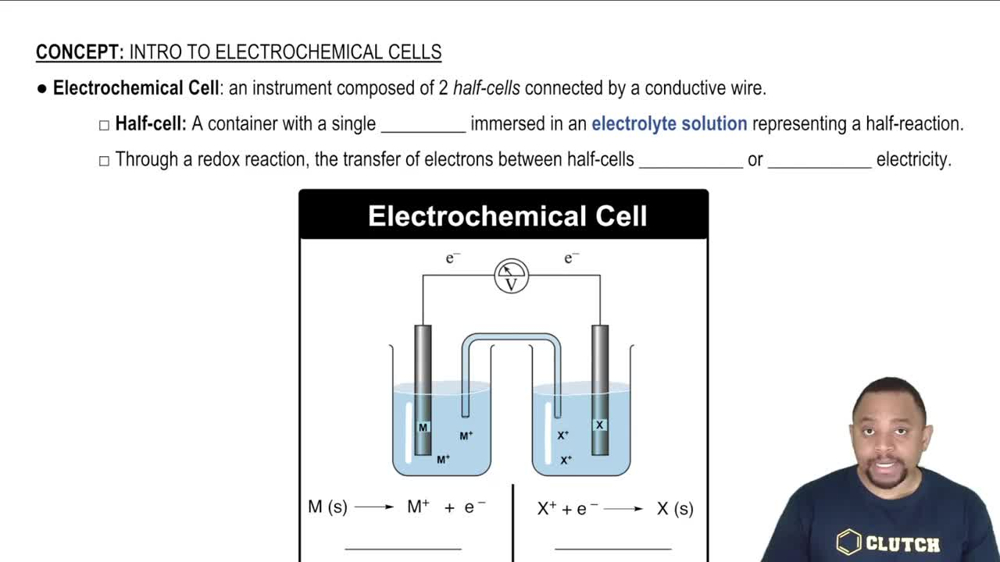
Electrochemical Cell and EMF
Balancing Redox Reactions

Using standard reduction potentials (Appendix E), calculate the standard emf for each of the following reactions: (a) Cl21g2 + 2 I-1aq2 ¡ 2 Cl-1aq2 + I21s2
Using standard reduction potentials (Appendix E), calculate the standard emf for each of the following reactions: (b) Ni1s2 + 2 Ce4+1aq2 ¡ Ni2+1aq2 + 2 Ce3+1aq2
Using standard reduction potentials (Appendix E), calculate the standard emf for each of the following reactions: (d) 2 NO3-1aq2 + 8 H+1aq2 + 3 Cu1s2 ¡ 2 NO1g2 + 4 H2O1l2 + 3 Cu2+1aq2
The standard reduction potentials of the following halfreactions are given in Appendix E:
Ag+(aq) + e- → Ag(s)
Cu2+(aq) + 2 e- → Cu(s)
Ni2+(aq) + 2 e- → Ni(s)
Cr3+(aq) + 3 e- → Cr(s)
(a) Determine which combination of these half-cell reactions leads to the cell reaction with the largest positive cell potential and calculate the value.
(b) Determine which combination of these half-cell reactions leads to the cell reaction with the smallest positive cell potential and calculate the value.
