16. Chemical Equilibrium
ICE Charts
Problem 91
Textbook Question
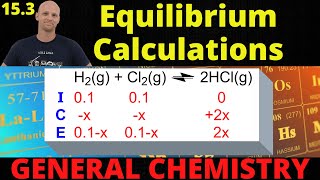
Textbook QuestionCarbon monoxide and chlorine gas react to form phosgene: CO(g) + Cl2(g) ⇌ COCl2(g) Kp = 3.10 at 700 K If a reaction mixture initially contains 215 torr of CO and 245 torr of Cl2, what is the mole fraction of COCl2 when equilibrium is reached?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
3636
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos