22. Organic Chemistry
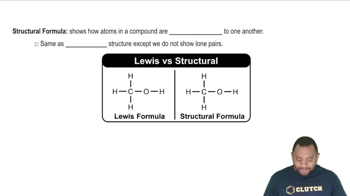
Structural Formula
Problem 2.142
Textbook Question
Textbook QuestionButane, the fuel used in disposable lighters, has the formula C4H10. The carbon atoms are connected in the sequence C-C-C-C, and each carbon has four covalent bonds. Draw the structural formula of butane.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
27
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos