8. Thermochemistry
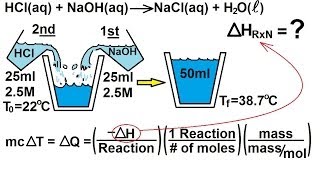
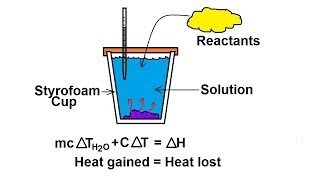
Constant-Pressure Calorimetry
Problem 55b
Textbook Question
Textbook QuestionWhen an 18.6-g sample of solid potassium hydroxide dissolves in 200.0 g of water in a coffee-cup calorimeter (Figure 5.18), the temperature rises from 23.7 to 44.5 °C. (b) Using your result from part (a), calculate H (in kJ>mol KOH) for the solution process. Assume that the specific heat of the solution is the same as that of pure water.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
10mPlay a video:
518
views
1
comments
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos