19. Chemical Thermodynamics
Entropy
Problem 44
Textbook Question
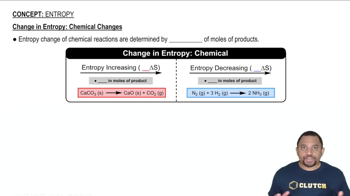
Textbook QuestionPredict the sign of the entropy change in the system for each of the following processes. (a) A solid sublimes. (b) A liquid freezes. (c) AgI precipitates from a solution containing Ag+ and I- ions. (d) Gaseous CO2 bubbles out of a carbonated beverage.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
592
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos