17. Acid and Base Equilibrium
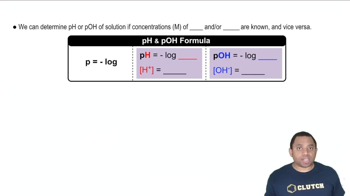
The pH Scale
Multiple Choice
Multiple ChoiceWhich of the following statement(s) on aqueous solutions is/are correct?
a) aqueous solutions have a pH of 7
b) as concentration of hydronium ion increases, concentration of hydroxide ion decreases
c) solutions of weaker acids generally have a higher pOH then solutions of stronger acids
d) pH of pure water equals to 7 at 35º C.
A
aqueous solutions have a pH of 7
B
as concentration of hydronium ion increases, concentration of hydroxide ion decreases
C
solutions of weaker acids generally have a higher pOH then solutions of stronger acids
D
pH of pure water equals to 7 at 35º C.
255
views
2
rank
1
comments
Related Videos
Related Practice
Showing 1 of 10 videos