21. Kinetic Theory of Ideal Gases
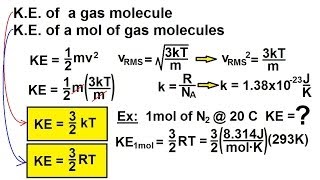
Average Kinetic Energy of Gases
Problem 37.96
Textbook Question
Textbook QuestionA rubidium atom (m = 85 u) is at rest with one electron in an excited energy level. When the electron jumps to the ground state, the atom emits a photon of wavelength ⋋ = 780 nm. (b) The recoil speed sets the lower limit on the temperature to which an ideal gas of rubidium atoms can be cooled in a laser-based atom trap. Using the kinetic theory of gases (Chapter 18), estimate this “lowest achievable” temperature.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
12
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos