In this page, we're going to discuss another product that forms when carbonyls react with alcohols, and that's called acetals. In general, I'm just going to say a few facts, and then we're going to go straight into the mechanism. The first fact I want you to know is that acetals, once formed, are actually stable in base. If you want to keep an acetal for a long time, keep it in a neutral to basic solution. However, they're easily hydrolyzed back to carbonyls using acid. That makes sense, guys, because remember that this is a reversible reaction. It's acid catalyzed. It makes sense that if you use acid, you're going to go back to the carbonyl and it's going to be in equilibrium. If you want to specifically make an acetal that not only has ROR but is actually cyclic, meaning it forms a ring, then you're going to have to use a diol because a diol is going to have carbons in the middle that are going to link together. For example, the diol that I would need here would be 1,2-ethanediol
21. Aldehydes and Ketones: Nucleophilic Addition
Acetal
21. Aldehydes and Ketones: Nucleophilic Addition
Acetal - Online Tutor, Practice Problems & Exam Prep
1
concept
General Mechanism
Video duration:
6mPlay a video:
Video transcript
2
Problem
ProblemProvide the chemical steps necessary for the following synthesis.

Video duration:
1mPlay a video:
Was this helpful?
Problem Transcript
3
Problem
ProblemProvide the chemical steps necessary for the following synthesis.
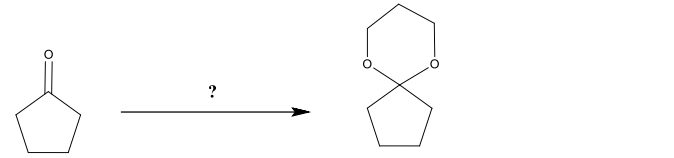
Video duration:
1mPlay a video:
Was this helpful?
Problem Transcript
4
Problem
ProblemDetermine the starting materials based on the acetal group present.

Video duration:
2mPlay a video:
Was this helpful?
Problem Transcript
Do you want more practice?
More setsYour Organic Chemistry tutors
Additional resources for Acetal
PRACTICE PROBLEMS AND ACTIVITIES (18)
- (•) For each of the following reactions, identify the bonds that are broken and formed. Be sure to indicate wh...
- What are the products of the following reactions? e.
- Draw the products of the following reactions: b.
- Biochemists studying the structure of collagen (a fibrous protein in connective tissue) found cross-links con...
- Propose mechanisms for the following reactions.(a) <IMAGE of reaction>
- Draw structures of the following derivatives.(e) acetaldehyde dimethyl acetal
- Predict the products formed when cyclohexanone reacts with the following reagents.(b) excess CH3OH, H+
- Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.(d) excess ethanol...
- Alcohols combine with ketones and aldehydes to form interesting derivatives, which we will discuss in Chapter ...
- For each compound,1. name the functional group.2. show what compound(s) result from complete hydrolysis.(d) &l...
- For each compound,1. name the functional group.2. show what compound(s) result from complete hydrolysis.(e) &l...
- There are three dioxane isomers: 1,2-dioxane, 1,3-dioxane, and 1,4-dioxane. One of these acts like an ether an...
- There are three dioxane isomers: 1,2-dioxane, 1,3-dioxane, and 1,4-dioxane. One of these acts like an ether an...
- (a) Simple aminoacetals hydrolyze quickly and easily in dilute acid. Propose a mechanism for hydrolysis of the...
- Hydration of an aldehyde is also catalyzed by hydroxide ion. Propose a mechanism for the reaction.
- Which of the following are a. hemiacetals? b. acetals? c. hydrates? 1. (CH3)2COCH3(OH)
- Which of the following are a. hemiacetals? b. acetals? c. hydrates? 3. (OCH3)2CH3CH
- Identify A through O:

