Hey, everyone. So in this video, we're going to take a look at Triacylglycerol Molecules. Now, here before that, we're going to say that our glycerol lipids, well, these are just lipids with fatty acid chains attached to a glycerol backbone. If we take a look at our lipids, remember we have hydrolyzable and also non-hydrolyzable portions of this. Here, we're talking about our Glycerolipids. We have waxes and sphingolipids, but we're only paying attention to our Glycerolipids here. Glycerol lipids can be broken down further into our Triacylglycerols and then we have our Phosphoglycerides. When we're talking about our Triacylglycerols, also known as Triglycerides, we can see that they have a glycerol backbone, and we have 3 fatty acids. And they're attached to the glycerol through ester bonds, so ester linkages. Now, fatty acids can all vary. All three fatty acids, they don't have to be the same. They can be, but they can also be different from one another. Now, here if we take a look, we're going to say if our C2 carbon, so this carbon here, it could be chiral depending on the fatty acids at carbon 1 and carbon 3. If we take a look here, we have our chiral center. It's connected to an H. It's connected to this portion here. And then, if we look, this group up here is different from this group down here. Because of that, carbon number 2 is chiral. Now, here if we go further, we can say that Triacylglycerols, they can function as a source of energy and storage, and they store in the form of fatty tissue otherwise known as adipose tissue. And this is happening within animals. So, just remember, we're talking about lipids, we're still looking at our hydrolyzable lipids. We're paying closer attention to our glycerol lipids which break down into our Triacylglycerols as well as our Phosphoglycerides. For Phosphoglycerides, it's almost the same except instead of having 3 fatty acid chains, we only have 2 with the third one being replaced with a phosphate group and an amino alcohol. Alright. So just keep this in mind when we're talking about our Triacylglycerol Molecules.
Triacylglycerols - Online Tutor, Practice Problems & Exam Prep
Triacylglycerols Concept 1
Video transcript
Triacylglycerols Example 1
Video transcript
It says to draw a triglyceride structure composed of palmitoleic acid for the first carbon, myristic acid for carbon 2, and oleic acid for carbon 3. Now, based on our memory tools, we know that myristic acid is a saturated fatty acid. It has 14 carbons and no pi bonds. Palmitoleic acid has 16 carbons and 1 pi bond, and oleic acid has 18 carbons and 1 pi bond. Additionally, their 1 pi bonds start on carbon 9. This will become important when drawing these structures.
Now, here we're going to say, step 1 is to draw the glycerol molecule and the 3 fatty acids. We're going to place OH groups next to the carboxyl groups of the fatty acid. So let's do that part first. Here we're gonna draw our glycerol molecule. We're gonna say we have CH2, CH2, CH2, and then we have our OH groups. Now we have to draw our fatty acids. Remember, our first one is an unsaturated fatty acid. We're gonna say it has 16 carbons, so 2, 4, 6, 8. Remember the first pi bond happens on carbon 9. So, 9, 10, 11, 12, 13, 14, 15, 16. Next, we have our myristic acid. It's saturated so it has 14 carbons and no pi bonds. So 2, 4, 6, 8, 10, 12, 14. And then finally, oleic acid has 18 carbons. 2, 4, 6, 8, 9 is where we have our double bond, 10, 11, 12, 13, 14, 15, 16, 17, 18. Now instead of OH on glycerol, we're just gonna write an O. So, we're gonna take away these H's here. And do not draw OH on the fatty acids.
So now it's up to us to connect for step 2. We're gonna form ester bonds between the glycerol OH groups and the 3 fatty acids. We're gonna connect the oxygens of the glycerol to the carbonyl carbons of the fatty acids. Doing this will give us our triglyceride. So, we're gonna have our CH2, and then CH2, CH2. Then we have O, O, and then O. Here's our ester linkage. So then we're gonna draw. Remember, we need 16 carbons. So 2, 4, 6, 8, 9, 10, 11, 12, 13, 14, 15, 16. Here, we need 14 carbons. 2, 4, 6, 8, 10, 12, 14. And then here we need 18 carbons. 2, 4, 6, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18 carbons. So, this will represent our triacylglycerol molecule or our triglyceride structure. This would be our final answer.
Triacylglycerols Concept 2
Video transcript
So in our continued discussion of Triacylglycerol Molecules, we now take a look at fats and oils. Now fats and oils can be seen as mixtures of different Triacylglycerols. Here, we're going to say that saturated fatty acids pack more tightly in a solid phase, which is going to help increase interactions between one another. These greater interactions are going to lead to an increase in the intermolecular forces between them, resulting in stronger intermolecular forces. And we're going to say because of this greater increase in intermolecular forces between these nearby molecules, this would translate into higher melting points. Now, if we take a look here at fats and oils, we have fats in terms of animal fats and then we have oils in terms of vegetables. With this, we can talk about melting point, saturation, as well as examples. Now with fats from animals, we're going to say here that the melting point tends to be higher and we're going to say they tend to be solids at room temperature. In terms of saturation, we're going to say that the number of double bonds tends to be low, and then we're going to say it's low in unsaturated fatty acids. A good example here, we have our glycerol backbone, and we have our 3 fatty acids. And we can see only 1 of the 3 has a pi bond involved, indicating very low levels of unsaturation. With oils, and here we're talking about in terms of vegetables, we're going to say they tend to have a low melting point and they tend to be liquids at room temperature. Here, we'd expect to have a higher number of pi bonds and a higher amount of unsaturation from fatty acids. If we take a look here at this example, we still have 3 fatty acid chains, but we can see with these fatty acid chains there's more presence of pi bonds, indicating greater levels of unsaturation. Again, this would mean that these fatty acids would not be able to pack as tightly around one another, so there's greater distance between each other which results in lower interactions, lower intermolecular forces because of lack of interactions, and therefore, a lower overall melting point. Right? So just keep that in mind when we're talking about fats and oils, and we're talking about levels of interactions and how that affects our overall melting point.
Triacylglycerols Example 2
Video transcript
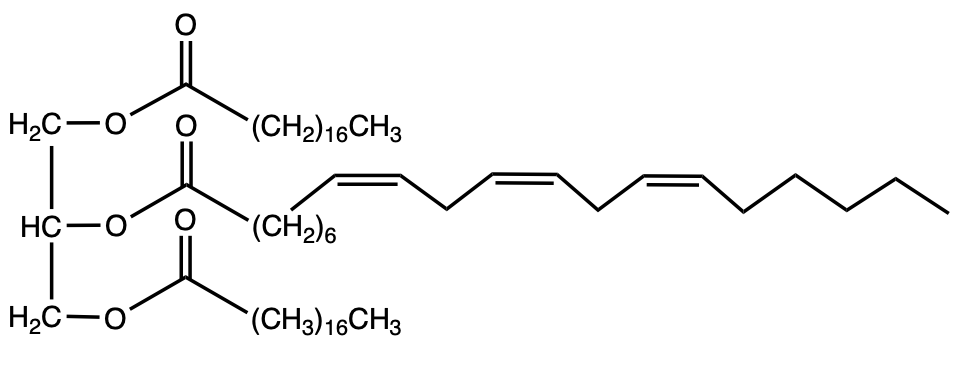
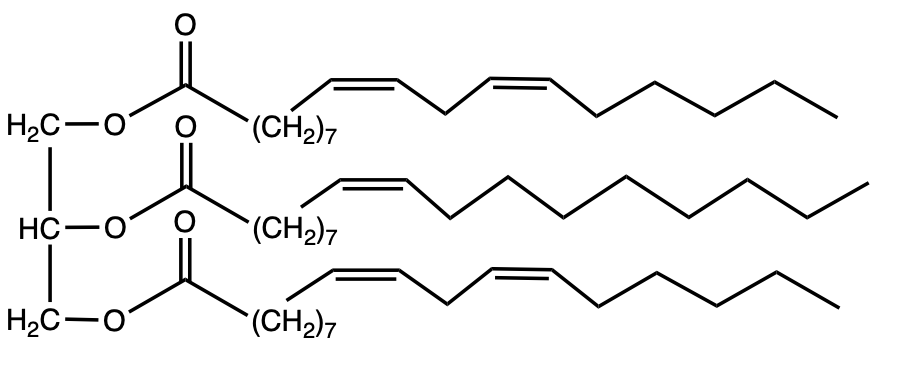
This example question asks, "Which Triacylglycerol would you expect to be liquid at room temperature?" Remember, we have fats, which are indicative of animals, and oils, which are indicative of vegetables and plants. Remember, the more double bonds or π bonds that we possess, the lower our melting point will be, and therefore it is more likely to exist as a liquid at room temperature. If we take a look at option a, we see that we have 1, 2, 3 double bonds or π bonds involved. And if we look at option b, we only have 1 π bond involved. The one most likely to be a liquid at room temperature would have to be option a. It possesses more double bonds, which will result in more kinking of the long fatty acid chain, which will result in less stacking of these structures on top of each other. Meaning they will exist more as a liquid at room temperature and have a lower melting point as a result. All right. Option b is less likely to be a liquid when compared to option a because it has much fewer π bonds or double bonds involved. So, again, in this particular question, option a is more likely to be a liquid than option b because of the presence of these π bonds or double bonds.
Draw a skeletal structure of a triglyceride with linolenic acid (C1) and 2 palmitoleic acids. State whether it would have high or low melting point.
Problem Transcript
Which triacylglycerol is optically active?