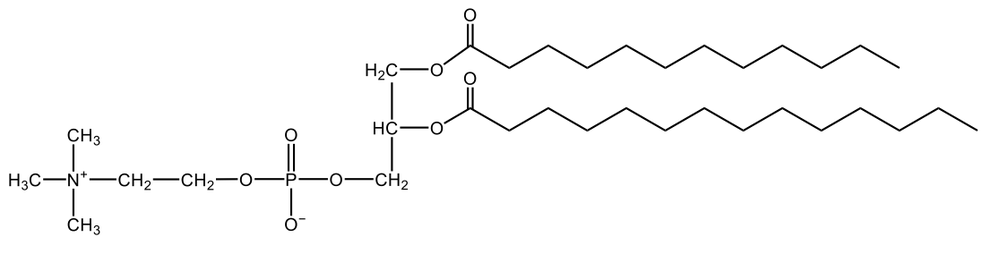
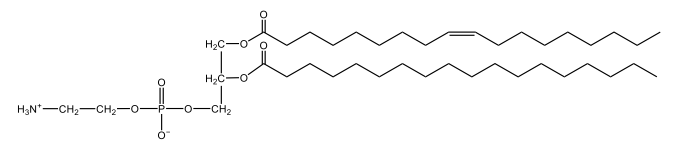
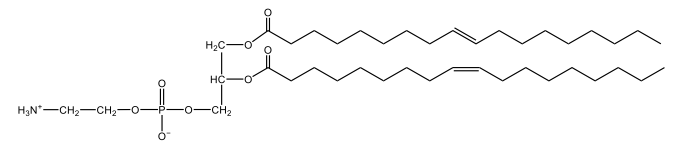
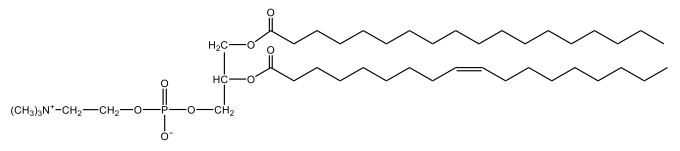
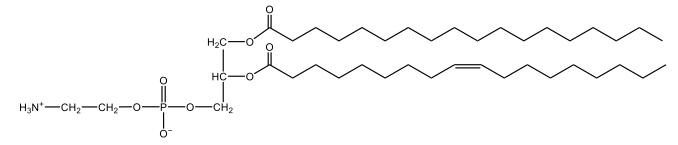
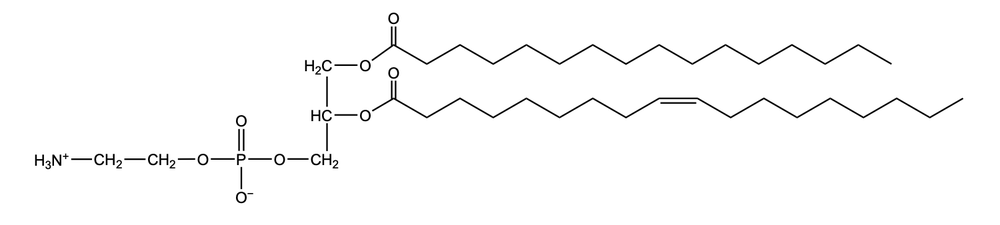
Phosphoglycerides are a specific type of phospholipid, which are lipids characterized by the presence of a phosphate group attached to a glycerol or sphingosine backbone. These molecules are amphipathic, meaning they possess both polar (hydrophilic) and non-polar (hydrophobic) regions. The hydrophilic head interacts favorably with water, while the hydrophobic tail repels it, allowing phospholipids to arrange themselves into a lipid bilayer, a fundamental structure of cell membranes.
Phospholipids can be categorized into hydrolyzable and non-hydrolyzable types, with phosphoglycerides falling under the hydrolyzable category. This group includes glycerol lipids and sphingolipids. Phosphoglycerides specifically consist of a glycerol backbone, two fatty acid chains, and a phosphate group linked to an amino alcohol. In contrast, sphingomyelin, another type of phospholipid, has a sphingosine backbone instead of glycerol, along with a fatty acid and a phosphate group connected to an amino alcohol.
These phospholipids are crucial components of all cell membranes, contributing to their structure and function. The rigidity and fluidity of the lipid bilayer are influenced by the types of fatty acids present in the phospholipids, highlighting their importance in biological systems. Understanding the structure and function of phosphoglycerides is essential for grasping the complexities of cellular membranes and their roles in various physiological processes.