- Download the worksheet to save time writing
- Start solving the practice problems
- If you're stuck, watch the video solutions
- See your summary to get more insights

What product is formed in the Heck reaction shown below?

Provide the starting materials required to produce the given compound using a Heck reaction.

Identify the alkenes that can be used in the following Heck reactions.

Show how the Suzuki reaction can be used to synthesize the compound shown below.

Draw the product obtained from the reaction shown below.

Determine the aryl halide that can be used to prepare the compound below, using the given alkenyl organoboron compound.

The Stille reaction bears a resemblance to the Suzuki reaction. It uses alkenyl-organotin in place of alkenyl-organoboron. This alkenyl-organotin compound can have a cis configuration, unlike alkenyl-organoboron, which only has a trans configuration. With the Stille reaction shown below, draw the structure of its resulting product.
Determine the corresponding product for the Stille coupling reaction shown below.
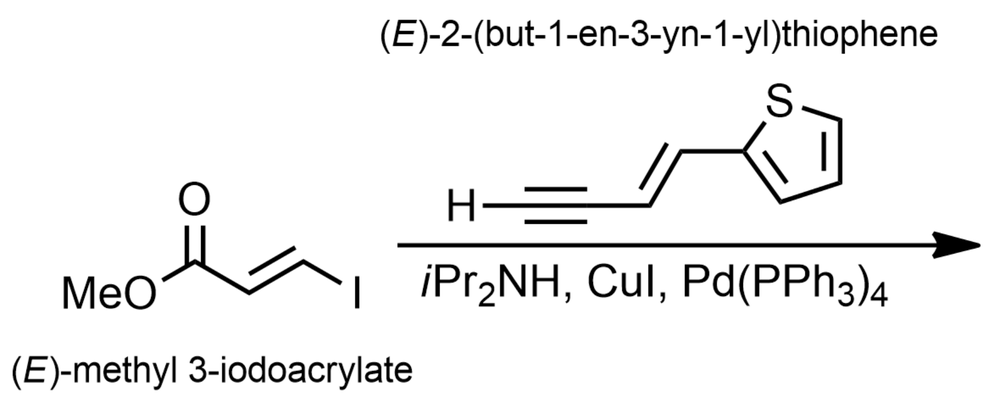
Propose the plausible product(s) of the Sonogashira coupling reaction between (E)-methyl 3-iodoacrylate and (E)-2-(but-1-en-3-yn-1-yl)thiophene.
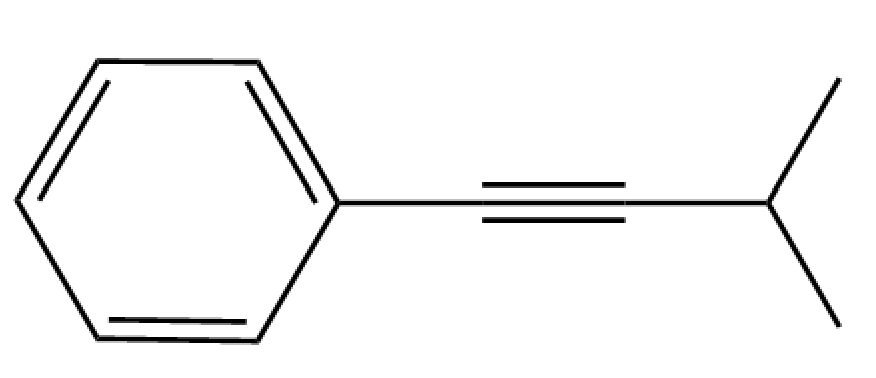
The Sonogashira reaction involves the coupling of an alkyne with an aryl halide using a palladium catalyst and triethylamine. Draw the initial reactants employed in the synthesis of the Sonogashira product shown below.
Draw the expected organic product of the Negishi coupling reaction shown below.

Provide the organozinc halide and organohalide used to make the compound below. The bond formed between the two components is indicated by the arrow. Consider the two possible combinations.

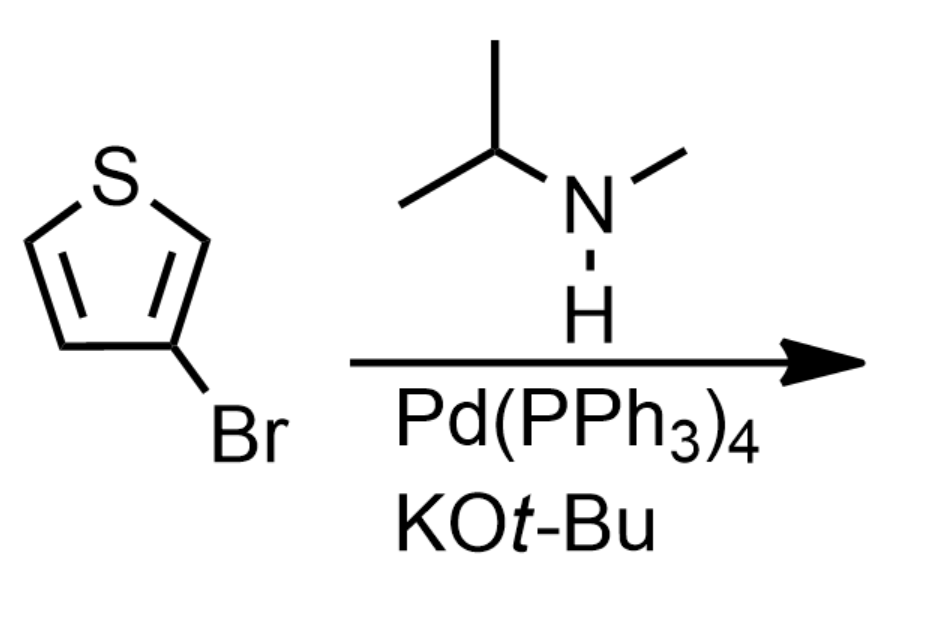
What is the product of the reaction below?
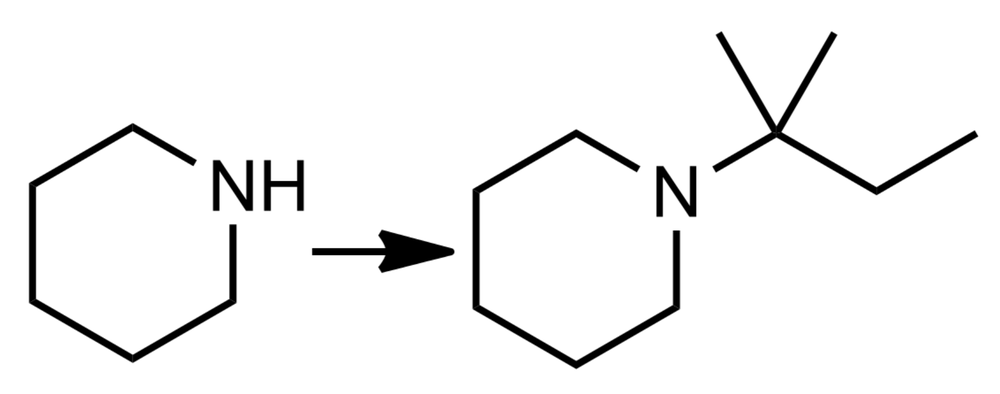
Suggest a method for synthesizing the amine on the right from the given reactant.