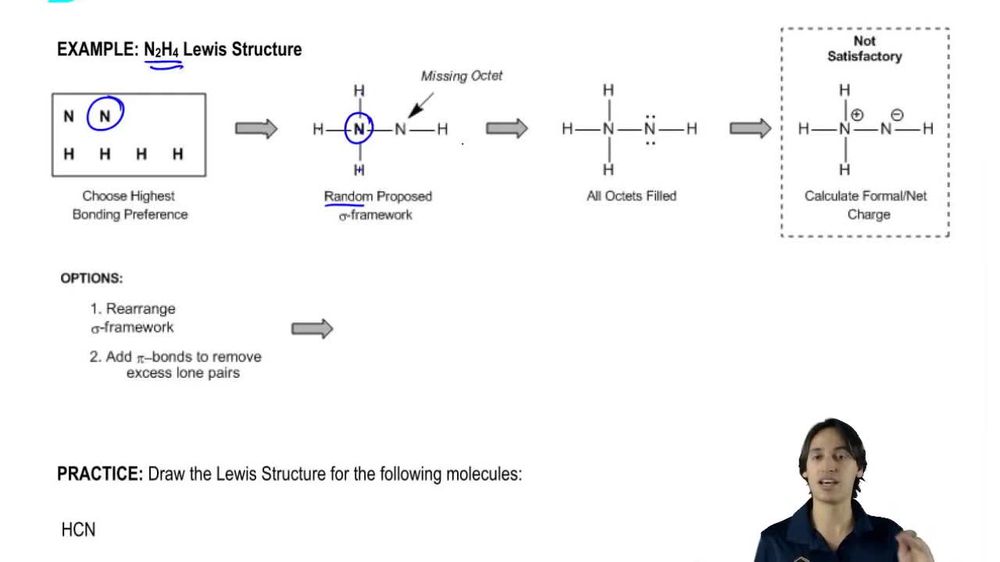
Write Lewis structures for the following molecular formulas. Circle any lone pairs (pairs of nonbonding electrons) in the structures.
i. C3H6 (one double bond)
j. C3H4 (two double bonds)
k. C3H4 (one triple bond)
 Verified step by step guidance
Verified step by step guidance Verified video answer for a similar problem:
Verified video answer for a similar problem: