3. Chemical Principles of Microbiology
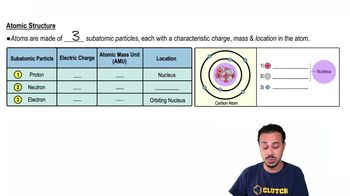
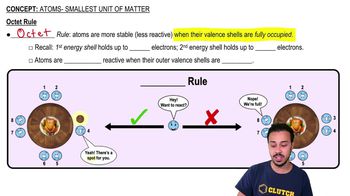
Atoms- Smallest Unit of Matter
Practice this topic
- Multiple Choice
A proton ___________:
a) Has one positive charge.
b) Has one AMU.
c) Is found in the nucleus of the atom.
d) Only a and b are true.
e) a, b, and c are true.
9329views237rank - Multiple Choice
The average oxygen atom has a mass number of 16 and an atomic number of 8. This means that the number of neutrons in this oxygen atom is:
a) 24.
b) 8.
c) 16.
d) 4.
e) 2.
11074views160rank - Multiple Choice
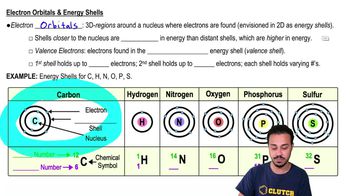
How many valence electrons does an atom with five total electrons have?
a) 5.
b) 7.
c) 3.
d) 2.
e) 1.
8049views127rank - Multiple Choice
Which of the following is true about electron energy shells?
a) They represent regions around the nucleus in which the electrons orbit.
b) The shells closest to the nucleus contain electrons with higher energy.
c) They contain electrons of the same energy.
d) a and b only.
e) a and c only.
7335views126rank - Textbook Question
What is a chemical element?
923views - Textbook Question
Diagram the electronic configuration of a carbon atom.
957views - Textbook Question
The term trace elements refers to
a. The elements CHONPS.
b. Vitamins.
c. Nitrogen, phosphorus, and sulfur.
d. Small mineral requirements.
e. Toxic substances.
753views - Textbook Question
Using the periodic table, answer the following questions:
a. What is the atomic number for lithium (Li)?
b. What is the atomic mass of oxygen (O)?
c. What is the chemical symbol for potassium?
d. How many protons does nitrogen have?
e. How many neutrons does lead have?
741views