- Download the worksheet to save time writing
- Start solving the practice problems
- If you're stuck, watch the video solutions
- See your summary to get more insights

What is the classification of the given amine (primary, secondary, or tertiary)?
CH3(CH2)4NHCH3
Consider the structure shown below:

Determine the amine classification of each nitrogen atom as either heterocyclic aromatic, primary, secondary, tertiary, or quaternary.
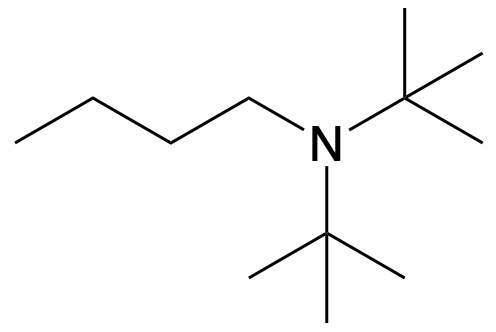
Determine whether the following compound is classified as a primary, secondary, or tertiary amine.
What is the corresponding structure of the following?
N-ethylhexanamine
Provide the name of the given compound:
(CH3CH2CH2CH2CH2CH2)2NH
Consider the amine below:
Provide its common name and classify it as primary (1°), secondary (2°), or tertiary (3°) amine.
Provide two possible structures for dicyclohexylamine hydrochloride and give its name as an ammonium salt.

Consider the ammonium salt shown below. Provide its name and identify if it is a salt of primary, secondary, or tertiary amine.

What is the condensed structural formula of N-ethyl-N-propylpentylammonium bromide? Is it a salt of a primary, secondary, or tertiary amine?
Provide the product of the reaction below (Note: Solution may involve concepts learned from different chapters):

Aniline reacts with HCl and water under different conditions. Provide the balanced chemical equations for the following reactions involving aniline:
- Its reaction with water.
- Its neutralization with HCl.
Determine if the molecule shown below is soluble in water. Explain your answer.