8. Gases, Liquids and Solids
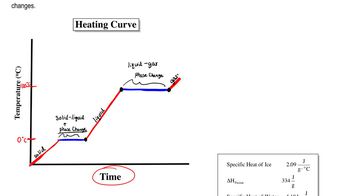
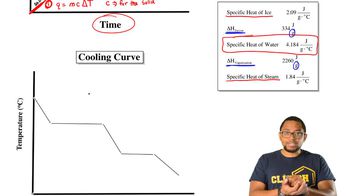
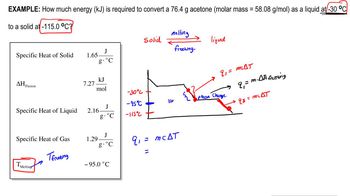
Heating and Cooling Curves
Problem 53
Textbook Question
Textbook QuestionUsing the values for the heat of fusion, specific heat of water, and/or heat of vaporization, calculate the amount of heat energy in each of the following: c. kilojoules needed to melt 24.0 g of ice at 0 °C, warm the liquid to 100 °C, and change it to steam at 100 °C
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
12mPlay a video:
474
views
Was this helpful?
Related Videos
Related Practice