Which outer electron configurations would you expect to belong to a metalloid? ns2 b. ns2np6 c. ns2np5 d. ns2np2
Which experience a greater effective nuclear charge: the valence electrons in beryllium or the valence electrons in nitrogen? Why?
 Verified step by step guidance
Verified step by step guidance
Verified Solution
Key Concepts
Effective Nuclear Charge (Z_eff)

Shielding Effect
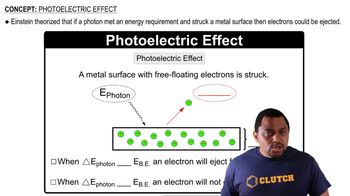
Trends in the Periodic Table

According to Coulomb's law, which pair of charged particles has the lowest potential energy? a. a particle with a 1- charge separated by 150 pm from a particle with a 2+ charge b. a particle with a 1- charge separated by 150 pm from a particle with a 1+ charge c. a particle with a 1- charge separated by 100 pm from a particle with a 3+ charge
According to Coulomb's law, rank the interactions between charged particles from lowest potential energy to highest potential energy. a. a 1+ charge and a 1- charge separated by 100 pm b. a 2+ charge and a 1- charge separated by 100 pm c. a 1+ charge and a 1+ charge separated by 100 pm d. a 1+ charge and a 1- charge separated by 200 pm
Arrange the atoms according to decreasing effective nuclear charge experienced by their valence electrons: S, Mg, Al, Si.
If core electrons completely shielded valence electrons from nuclear charge (i.e., if each core electron reduced nuclear charge by 1 unit) and if valence electrons did not shield one another from nuclear charge at all, what would be the effective nuclear charge experienced by the valence electrons of each atom? a. K
If core electrons completely shielded valence electrons from nuclear charge (i.e., if each core electron reduced nuclear charge by 1 unit) and if valence electrons did not shield one another from nuclear charge at all, what would be the effective nuclear charge experienced by the valence electrons of each atom? b. Ca
