A quantity of N2 occupies a volume of 1.0 L at 300 K and 1.0 atm. The gas expands to a volume of 3.0 L as the result of a change in both temperature and pressure. Find the density of the gas at these new conditions.
A mixture of 8.0 g CH4 and 8.0 g Xe is placed in a container and the total pressure is found to be 0.44 atm. Determine the partial pressure of CH4.
 Verified step by step guidance
Verified step by step guidance
Verified Solution
Key Concepts
Dalton's Law of Partial Pressures

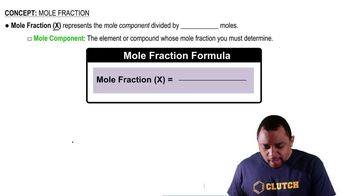
Mole Fraction
Ideal Gas Law

A mixture of CO(g) and O2(g) in a 1.0-L container at 1.0×103 K has a total pressure of 2.2 atm. After some time, the total pressure falls to 1.9 atm as the result of the formation of CO2. Determine the mass (in grams) of CO2 that forms.
The radius of a xenon atom is 1.3×10– 8 cm. A 100-mL flask is filled with Xe at a pressure of 1.0 atm and a temperature of 273 K. Calculate the fraction of the volume that is occupied by Xe atoms. (Hint: The atoms are spheres.)
Binary compounds of alkali metals and hydrogen react with water to liberate H2(g). The H2 from the reaction of a sample of NaH with an excess of water fills a volume of 0.490 L above the water. The temperature of the gas is 35 °C and the total pressure is 758 mmHg. Determine the mass of H2 liberated and the mass of NaH that reacted.
In a given diffusion apparatus, 15.0 mL of HBr gas diffuses in 1.0 min. In the same apparatus and under the same conditions, 20.3 mL of an unknown gas diffuses in 1.0 min. The unknown gas is a hydrocarbon. Find its molecular formula.
A sample of N2O3(g) has a pressure of 0.017 atm. The temperature (in K) is doubled and the N2O3 undergoes complete decomposition to NO2(g) and NO(g). Find the total pressure of the mixture of gases assuming constant volume and no additional temperature change.
