This reaction has an activation energy of zero in the gas phase: CH3 + CH3 → C2H6
a. Would you expect the rate of this reaction to change very much with temperature?
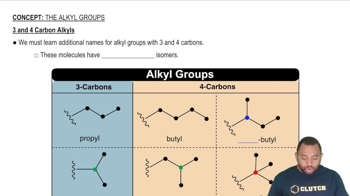
 Verified step by step guidance
Verified step by step guidance


This reaction has an activation energy of zero in the gas phase: CH3 + CH3 → C2H6
a. Would you expect the rate of this reaction to change very much with temperature?
Consider the two reactions:
O + N2 → NO + N Ea = 315 kJ/mol
Cl + H2 → HCl + H Ea = 23 kJ/mol
a. Why is the activation barrier for the first reaction so much higher than that for the second?
Consider the two reactions:
O + N2 → NO + N Ea = 315 kJ/mol
Cl + H2 → HCl + H Ea = 23 kJ/mol
b. The frequency factors for these two reactions are very close to each other in value. Assuming that they are the same, calculate the ratio of the reaction rate constants for these two reactions at 25 °C.
Consider the gas-phase reaction: H2(g) + I2(g) → 2 HI(g) The reaction was experimentally determined to be first order in H2 and first order in I2. Consider the proposed mechanisms. Proposed mechanism I: H2(g) + I2(g) → 2 HI(g) Single step Proposed mechanism II: I2(g) Δk1k-12 I(g) Fast H2( g) + 2 I( g) → k22 HI( g) Slow a. Show that both of the proposed mechanisms are valid.
Consider the gas-phase reaction: H2(g) + I2(g) → 2 HI(g) The reaction was experimentally determined to be first order in H2 and first order in I2. Consider the proposed mechanisms. Proposed mechanism I: H2(g) + I2(g) → 2 HI(g) Single step Proposed mechanism II: I2(g) Δk1k-12 I(g) Fast H2( g) + 2 I( g) → k22 HI( g) Slow b. What kind of experimental evidence might lead you to favor mechanism II over mechanism I?
Phosgene (Cl2CO), a poison gas used in World War I, is formed
by the reaction of Cl2 and CO. The proposed mechanism for the
reaction is:
Cl2Δ2 Cl (fast, equilibrium)
Cl + COΔClCO (fast, equilibrium)
ClCO + Cl2¡Cl2CO + Cl (slow)