Textbook Question
Use the Born–Haber cycle and data from Appendix IIB, Chapter 9 and this chapter to calculate the lattice energy of KCl. (ΔHsub for potassium is 89.0 kJ>mol.)
 Verified step by step guidance
Verified step by step guidance

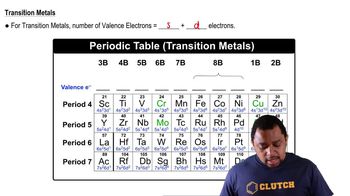
Use the Born–Haber cycle and data from Appendix IIB, Chapter 9 and this chapter to calculate the lattice energy of KCl. (ΔHsub for potassium is 89.0 kJ>mol.)
Use the Born–Haber cycle and data from Appendix IIB and Table 10.3 to calculate the lattice energy of CaO. (ΔHsub for calcium is 178 kJ>mol; IE1 and IE2 for calcium are 590 kJ>mol and 1145 kJ>mol, respectively; EA1 and EA2 for O are -141 kJ>mol and 744 kJ>mol, respectively.)
Write the Lewis structure for each molecule. a. PH3 b. SCl2 c. HI
Write the Lewis structure for each molecule. a. SF2
Write the Lewis structure for each molecule. b. SiH4
Write the Lewis structure for each molecule. d. CH3SH (C and S central)