Write a Lewis structure that obeys the octet rule for each molecule or ion. Include resonance structures if necessary and assign formal charges to each atom. a. SeO2
Ch.10 - Chemical Bonding I: The Lewis Model
Chapter 10, Problem 67
How important is the resonance structure shown here to the overall structure of carbon dioxide? Explain.


Verified Solution
Video duration:
5mWas this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Resonance Structures
Resonance structures are different Lewis structures for the same molecule that depict the same arrangement of atoms but differ in the distribution of electrons. They are used to represent delocalized electrons within certain molecules, indicating that the actual structure is a hybrid of these forms. In carbon dioxide, resonance structures help illustrate the equivalent bonding between carbon and oxygen atoms, contributing to the molecule's stability.
Recommended video:
Guided course

Resonance Structures
Molecular Geometry
Molecular geometry refers to the three-dimensional arrangement of atoms in a molecule. In carbon dioxide, the linear geometry arises from the arrangement of the double bonds between carbon and oxygen, which minimizes electron pair repulsion according to VSEPR theory. Understanding the geometry is crucial for predicting the molecule's reactivity and interactions with other substances.
Recommended video:
Guided course
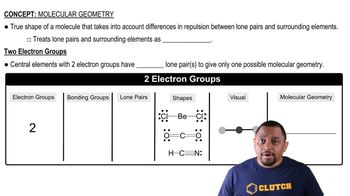
Molecular Geometry with Two Electron Groups
Bond Order
Bond order is a measure of the number of chemical bonds between a pair of atoms, which influences the bond strength and stability. In carbon dioxide, the bond order is 2 for each C=O bond, indicating a double bond. This concept is essential for understanding the overall stability and reactivity of the molecule, as higher bond orders typically correlate with stronger, shorter bonds.
Recommended video:
Guided course

Average Bond Order
Related Practice
Textbook Question
935
views
Textbook Question
Write a Lewis structure that obeys the octet rule for each ion. Include resonance structures if necessary and assign formal charges to each atom. a. ClO3- b. ClO4- c. NO3- d. NH4+
1632
views
Textbook Question
Use formal charges to identify the better Lewis structure.
1166
views
1
rank
Textbook Question
In N2O, nitrogen is the central atom and the oxygen atom is terminal. In OF2, however, oxygen is the central atom. Use formal charges to explain why.
1716
views
Textbook Question
Draw the Lewis structure (including resonance structures) for the acetate ion (CH3COO-). For each resonance structure, assign formal charges to all atoms that have formal charge.
1572
views
Textbook Question
Draw the Lewis structure (including resonance structures) for methyl azide (CH3N3). For each resonance structure, assign formal charges to all atoms that have formal charge.
2220
views
