Calculate the solubility (in grams per 1.00⨉102 mL of solution) of magnesium hydroxide in a solution buffered at pH = 10. How does this compare to the solubility of Mg(OH)2 in pure water?
Predict whether a precipitate will form if you mix 75.0 mL of a NaOH solution with pOH = 2.58 with 125.0 mL of a 0.018 M MgCl2 solution. Identify the precipitate, if any.

Verified Solution
Key Concepts
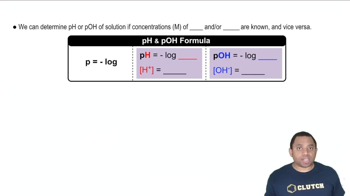
pOH and pH Relationship
Solubility Product Constant (Ksp)

Precipitation Reactions

Determine if each compound is more soluble in acidic solution than it is in pure water. Explain. a. BaCO3 b. CuS c. AgCl d. PbI2
A solution containing sodium fluoride is mixed with one containing calcium nitrate to form a solution that is 0.015 M in NaF and 0.010 M in Ca(NO3)2. Does a precipitate form in the mixed solution? If so, identify the precipitate.
Determine the minimum concentration of the precipitating agent on the right to cause precipitation of the cation from the solution on the left. a. 0.035 M Ba(NO3)2; NaF
Determine the minimum concentration of the precipitating agent on the right to cause precipitation of the cation from the solution on the left. b. 0.085 M CaI2; K2SO4
Determine the minimum concentration of the precipitating agent on the right to cause precipitation of the cation from the solution on the left. c. 0.0018 M AgNO3; RbCl
