A 1.20-g sample of dry ice is added to a 755 mL flask containing nitrogen gas at a temperature of 25.0 °C and a pressure of 725 mmHg. The dry ice sublimes (converts from solid to gas), and the mixture returns to 25.0 °C. What is the total pressure in the flask?
What is the mole fraction of oxygen gas in air (see Table 5.3)? What volume of air contains 10.0 g of oxygen gas at 273 K and 1.00 atm?
 Verified step by step guidance
Verified step by step guidance
Verified Solution
Key Concepts
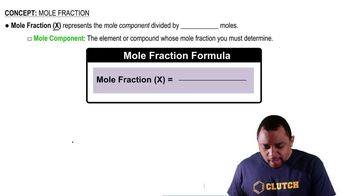
Mole Fraction
Ideal Gas Law

Standard Temperature and Pressure (STP)

A 275-mL flask contains pure helium at a pressure of 752 torr. A second flask with a volume of 475 mL contains pure argon at a pressure of 722 torr. If we connect the two flasks through a stopcock and we open the stopcock, what is the partial pressure of argon?
A gas mixture contains 1.25 g N2 and 0.85 g O2 in a 1.55 L container at 18 °C. Calculate the mole fraction and partial pressure of each component in the gas mixture.
The hydrogen gas formed in a chemical reaction is collected over water at 30.0 °C at a total pressure of 732 mmHg. What is the partial pressure of the hydrogen gas collected in this way? If the total volume of gas collected is 722 mL, what mass of hydrogen gas is collected?
The air in a bicycle tire is bubbled through water and collected at 25 °C. If the total volume of gas collected is 5.45 L at a temperature of 25 °C and a pressure of 745 torr, how many moles of gas were in the bicycle tire?
The zinc in a copper-plated penny will dissolve in hydrochloric acid if the copper coating is filed down in several spots (so that the hydrochloric acid can get to the zinc). The reaction between the acid and the zinc is 2 H+(aq) + Zn(s) → H2(g) + Zn2+(aq). When the zinc in a certain penny dissolves, the total volume of gas collected over water at 25 °C is 0.951 L at a total pressure of 748 mmHg. What mass of hydrogen gas is collected?
