Here are the essential concepts you must grasp in order to answer the question correctly.
Solubility Product Constant (Ksp)
The solubility product constant (Ksp) is an equilibrium constant that applies to the solubility of sparingly soluble ionic compounds. It quantifies the extent to which a compound can dissolve in water, represented by the concentrations of its ions at equilibrium. For the reaction involving FeS, Ksp helps determine the solubility of FeS in the presence of complexing agents like CN-.
Recommended video:
Solubility Product Constant
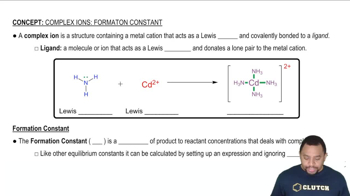
Formation Constant (Kf)
The formation constant (Kf) is a measure of the stability of a complex ion in solution. It quantifies the equilibrium between the free ions and the complex formed when they react. In the given reaction, Kf is crucial for understanding how effectively CN- ions bind with Fe2+ to form the complex ion Fe(CN)64-, influencing the overall equilibrium of the reaction.
Recommended video:
Complex Ions and Formation Constant
Equilibrium Constant (K)
The equilibrium constant (K) for a reaction is derived from the concentrations of the products and reactants at equilibrium. It provides insight into the extent of the reaction and can be calculated using Ksp and Kf values. In this case, the equilibrium constant for the reaction involving FeS and CN- can be determined by combining the Ksp of FeS and the Kf for the formation of the complex ion, reflecting the relationship between solubility and complexation.
Recommended video:
 Verified step by step guidance
Verified step by step guidance