Write balanced molecular and net ionic equations for the reactions of (c) hydrobromic acid with magnesium (d) acetic acid, CH3COOH, with zinc.
Using the activity series (Table 4.5), write balanced chemical equations for the following reactions. If no reaction occurs, write NR. (d) hydrogen gas is bubbled through an aqueous solution of nickel(II) chloride
 Verified step by step guidance
Verified step by step guidance
Verified Solution
Key Concepts
Activity Series
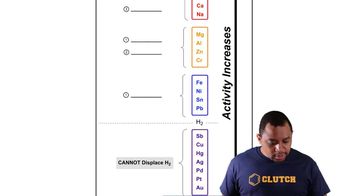
Redox Reactions

Solubility Rules

Using the activity series (Table 4.5), write balanced chemical equations for the following reactions. If no reaction occurs, write NR. (a) Iron metal is added to a solution of copper(II) nitrate (b) zinc metal is added to a solution of magnesium sulfate
Using the activity series (Table 4.5), write balanced chemical equations for the following reactions. If no reaction occurs, write NR. (c) hydrobromic acid is added to tin metal
Using the activity series (Table 4.5), write balanced chemical equations for the following reactions. If no reaction occurs, write NR. (e) aluminum metal is added to a solution of cobalt(II) sulfate.
Using the activity series (Table 4.5), write balanced chemical equations for the following reactions. If no reaction occurs, write NR. (a) Nickel metal is added to a solution of copper(II) nitrate
Using the activity series (Table 4.5), write balanced chemical equations for the following reactions. If no reaction occurs, write NR. (b) a solution of zinc nitrate is added to a solution of magnesium sulfate (c) hydrochloric acid is added to gold metal
