The reaction of MnO4– with oxalic acid (H2C2O4) in acidic solution, yielding Mn2+ and CO2 gas, is widely used to determine the concentration of permanganate solutions. (b) Use the data in Appendix D to calculate E° for the reaction. (c) Show that the reaction goes to completion by calculating the values of ∆G° and K at 25 °C. (H2C2O4) in acidic solution, yielding Mn2+ and CO2 gas, is widely used to determine the concentration of permanganate solutions.

A concentration cell has the same half-reactions at the anode and cathode, but a voltage results from different concentrations in the two electrode compartments.(b) A similar cell has 0.10 M Cu2+ in both compartments. When a stoichiometric amount of ethylenediamine (NH2CH2CH2NH2) is added to one compartment, the measured cell potential is 0.179 V. Calculate the formation constant Kf for the complex ion Cu(NH2CH2CH2CH2)22+. Assume there is no volume change.
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Concentration Cells

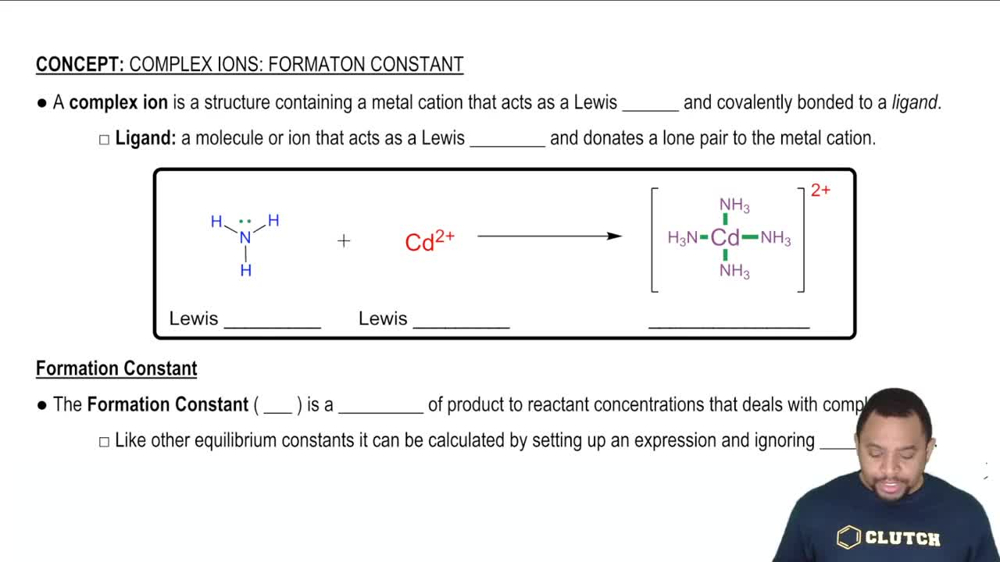
Formation Constant (Kf)
Nernst Equation

The reaction of MnO4– with oxalic acid (H2C2O4) in acidic solution, yielding Mn2+ and CO2 gas, is widely used to determine the concentration of permanganate solutions. (d) A 1.200 g sample of sodium oxalate (Na2C2O4) is dissolved in dilute H2SO4 and then titrated with a KMnO4 solution. If 32.50 mL of the KMnO4 solution is required to reach the equivalence point, what is the molarity of the KMnO4 solution?
Consider the redox titration (Section 4.13) of 120.0 mL of 0.100 M FeSO4 with 0.120 M K2Cr2O7 at 25 °C, assuming that the pH of the solution is maintained at 2.00 with a suitable buffer. The solution is in contact with a platinum electrode and constitutes one half-cell of an electrochemical cell. The other half-cell is a standard hydrogen electrode. The two half-cells are connected with a wire and a salt bridge, and the progress of the titration is monitored by measuring the cell potential with a voltmeter. (a) Write a balanced net ionic equation for the titration reaction, assuming that the products are Fe3+ and Cr3+.
