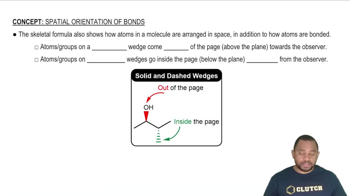
Textbook Question
Use the MO energy diagram in Figure 8.22b to describe thebonding in O2+, O2, and O2-. Which of the three is likely tobe stable? What is the bond order of each? Which containunpaired electrons?
991
views

 McMurry 8th Edition
McMurry 8th Edition Ch.8 - Covalent Compounds: Bonding Theories and Molecular Structure
Ch.8 - Covalent Compounds: Bonding Theories and Molecular Structure Problem 96
Problem 96 Verified step by step guidance
Verified step by step guidance