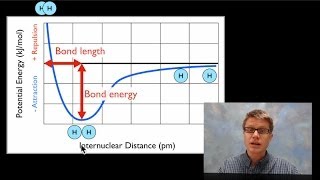
11. Bonding & Molecular Structure
Bond Energy
Problem 105b
Textbook Question
Textbook QuestionIf hydrogen were used as a fuel, it could be burned according to this reaction: H2( g) + 1 2 O2( g)¡H2O( g) Use average bond energies to calculate ΔHrxn for the combustion of methane (CH4).
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
8mPlay a video:
748
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos