Textbook Question
Like all equilibrium constants, the value of Kw depends on temperature. At body temperature (37 °C), Kw = 2.4×10–14. What are the [H3O+] and pH of pure water at body temperature?
4273
views
10
rank

 Verified step by step guidance
Verified step by step guidance

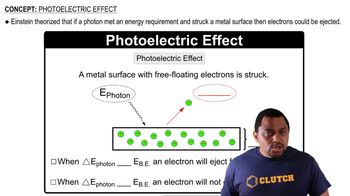

Like all equilibrium constants, the value of Kw depends on temperature. At body temperature (37 °C), Kw = 2.4×10–14. What are the [H3O+] and pH of pure water at body temperature?
Calculate [H3O+] and [OH–] for each solution at 25 °C. a. pH = 8.55
Calculate [H3O+] and [OH–] for each solution at 25 °C. b. pH = 11.23 c. pH = 2.87
Complete the table. (All solutions are at 25 °C.)
The value of Kw increases with increasing temperature. Is the autoionization of water endothermic or exothermic?