Textbook Question
Compare some of the physical properties of H2S, NaH, and PdHx.
84
views

 Verified step by step guidance
Verified step by step guidance

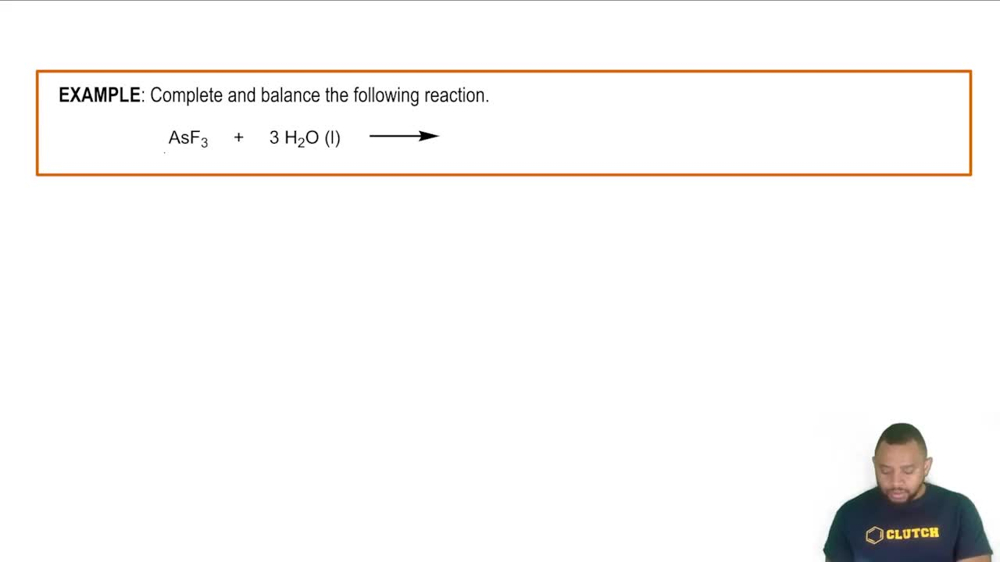

Compare some of the physical properties of H2S, NaH, and PdHx.
GeCl4 reacts with Cl- to give GeCl62-, but CCl4 does not react with excess Cl-. Explain.
The following models represent the structures of binary hydrides
of second-row elements:
(a) Identify the nonhydrogen atom in each case, and write the molecular formula for each hydride.
Which of the following oxides will be more soluble in acidic solution? (LO 22.14)
(a) SiO2 (b) NO2
(c) BaO (d) B2O3
Describe the structures of the white and red allotropes of phosphorus, and explain why white phosphorus is so reactive.
Explain why the properties of boron differ so markedly from the properties of the other group 3A elements.