Draw an MO energy diagram and predict the bond order of Be2+ and Be2- . Do you expect these molecules to exist in the gas phase?
 Tro 4th Edition
Tro 4th Edition Ch.10 - Chemical Bonding II: Molecular Shapes & Valence Bond Theory
Ch.10 - Chemical Bonding II: Molecular Shapes & Valence Bond Theory Problem 74
Problem 74Sketch the bonding and antibonding molecular orbitals that result from linear combinations of the 2pz atomic orbitals in a homonuclear diatomic molecule. (The 2pz orbitals are those whose lobes are oriented perpendicular to the bonding axis.) How do these molecular orbitals differ from those obtained from linear combinations of the 2py atomic orbitals? (The 2py orbitals are also oriented perpendicular to the bonding axis, but also perpendicular to the 2pz orbitals.)
 Verified step by step guidance
Verified step by step guidance
Verified Solution
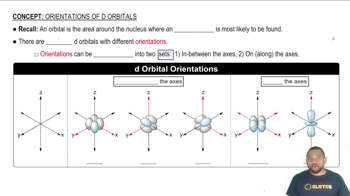
Key Concepts
Molecular Orbitals

Linear Combination of Atomic Orbitals (LCAO)

Orbital Orientation and Symmetry
Draw an MO energy diagram and predict the bond order of Li2+ and Li2-. Do you expect these molecules to exist in the gas phase?
Sketch the bonding and antibonding molecular orbitals that result from linear combinations of the 2px atomic orbitals in a homonuclear diatomic molecule. (The 2px orbitals are those whose lobes are oriented along the bonding axis.)
Using the molecular orbital energy ordering for second-row homonuclear diatomic molecules in which the π2p orbitals lie at lower energy than the σ2p, draw MO energy diagrams and predict the bond order in a molecule or ion with each number of total valence electrons. Will the molecule or ion be diamagnetic or paramagnetic? a. 4 b. 6
Using the molecular orbital energy ordering for second-row homonuclear diatomic molecules in which the π2p orbitals lie at lower energy than the σ2p, draw MO energy diagrams and predict the bond order in a molecule or ion with each number of total valence electrons. Will the molecule or ion be diamagnetic or paramagnetic?? b. 6 d. 9
Using the molecular orbital energy ordering for second-row homonuclear diatomic molecules in which the π2p orbitals lie at lower energy than the σ2p, draw MO energy diagrams and predict the bond order in a molecule or ion with each number of total valence electrons. Will the molecule or ion be diamagnetic or paramagnetic? c. 8