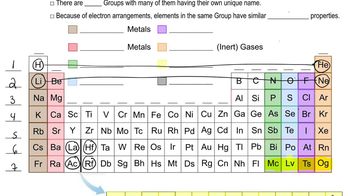
Group Trends in the Periodic Table
Elements in the same group of the periodic table exhibit similar chemical properties due to having the same number of valence electrons. For fourth-row elements, this includes elements like gallium, germanium, and arsenic. Recognizing these trends helps in predicting how an unknown element, represented as X in the question, might behave in chemical reactions, especially when forming compounds with halogens like bromine.


 Verified step by step guidance
Verified step by step guidance