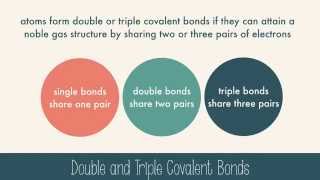
2. Chemistry
Covalent Bonds
2. Chemistry
Covalent Bonds
Additional 4 creators.
Learn with other creators
Showing 7 of 7 videos
Practice this topic
- Multiple Choice
When two atoms share a pair of electrons, the bonding is referred to as:
6891views101rank - Multiple Choice
What makes a covalent bond nonpolar?
5915views76rank6comments - Multiple Choice
If a covalent bond is polar:
6360views78rank - Multiple Choice
Bonds between two atoms that are equally or similarly electronegative are ________.
5979views62rank