23. The Second Law of Thermodynamics
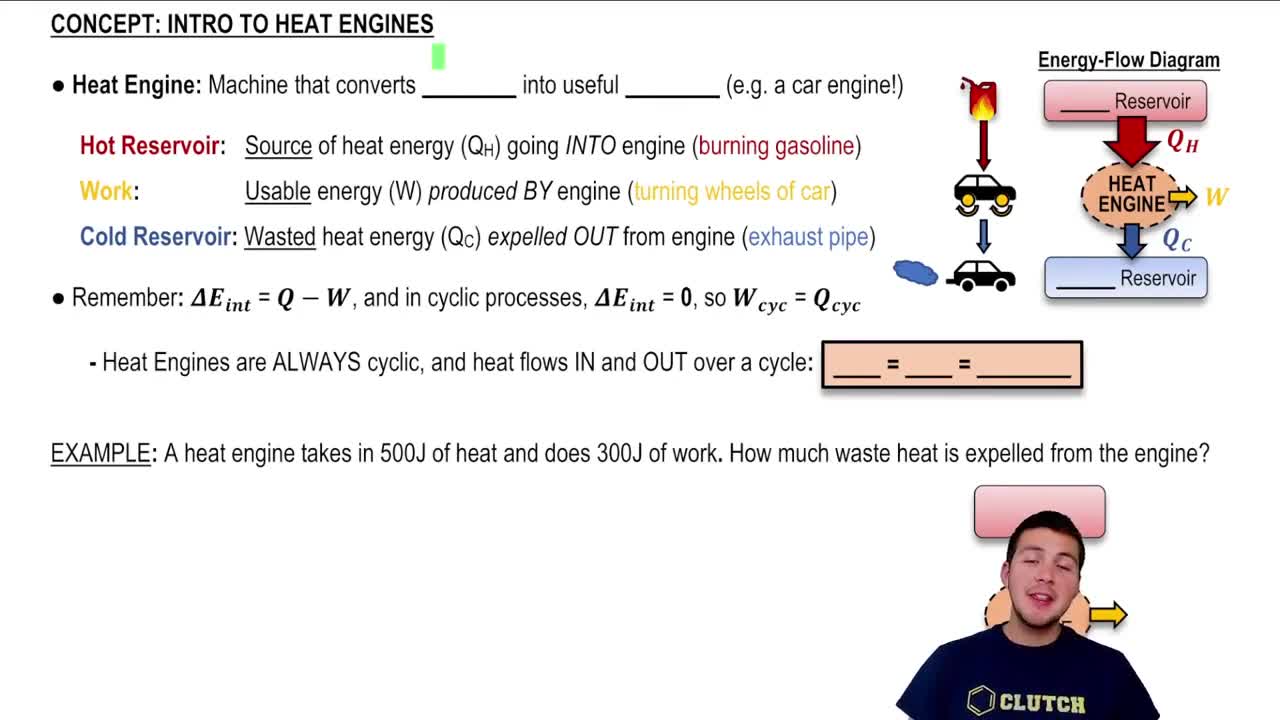
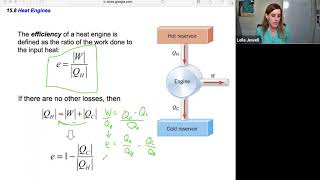
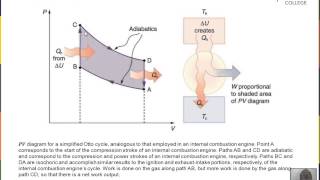
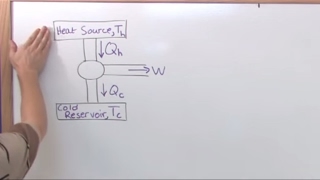
Heat Engines and the Second Law of Thermodynamics
Problem 20.15
Textbook Question
Textbook Question(II) Assume that a 65-kg hiker needs to eat 4.0 x 10³ kcal of energy to supply a day’s worth of metabolism ( = Q_H) . Estimate the elevation change the person can climb in one day, using only this amount of energy. As a fun and rough prediction, treat the person as an isolated heat engine, operating between the internal temperature of 37°C (98.6°F) and the ambient air temperature of 20°C.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
9mPlay a video:
49
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos