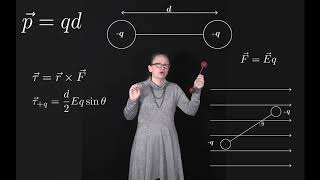24. Electric Force & Field; Gauss' Law
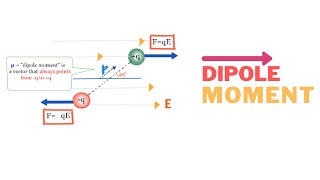
Dipole Moment
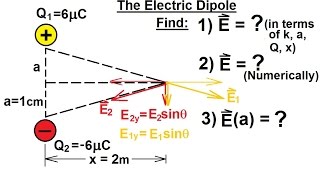
Problem 23b
Textbook Question
Textbook QuestionAn electric field can induce an electric dipole in a neutral atom or molecule by pushing the positive and negative charges in opposite directions. The dipole moment of an induced dipole is directly proportional to the electric field. That is, p⃗ =αE⃗ , where α is called the polarizability of the molecule. A bigger field stretches the molecule farther and causes a larger dipole moment. b. An ion with charge q is distance r from a molecule with polarizability α . Find an expression for the force Ē ion on dipole .
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
9mPlay a video:
151
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos