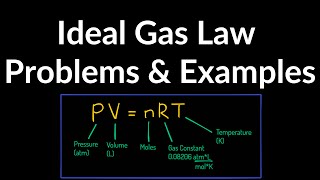
21. Kinetic Theory of Ideal Gases
The Ideal Gas Law
Problem 18.44a
Textbook Question
Textbook Question(III) How well does the ideal gas law describe the pressurized air in a scuba tank?
(a) To fill a typical scuba tank, an air compressor intakes about 2300 L of air at 1.0 atm and compresses this gas into the tank’s 12-L internal volume. If the filling process occurs at 20°C, show that a tank holds about 96 mol of air.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
141
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos