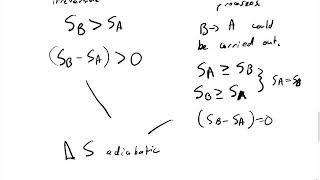23. The Second Law of Thermodynamics
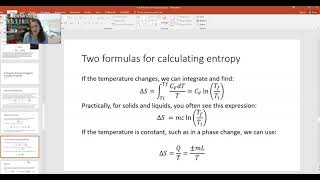
Entropy Equations for Special Processes
Problem 20.57b
Textbook Question
Textbook Question(III) A general theorem states that the amount of energy that becomes unavailable to do useful work in any process is equal to T_L∆S , where T_L is the lowest temperature available and ∆S is the total change in entropy during the process. Show that this is valid in the specific cases of:
(b) the free adiabatic expansion of an ideal gas;
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
37
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos