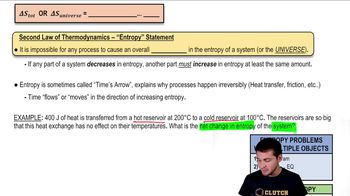
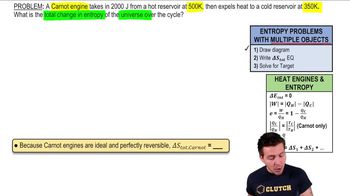
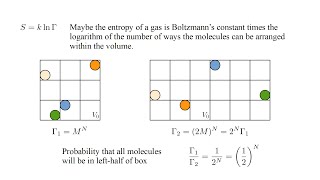
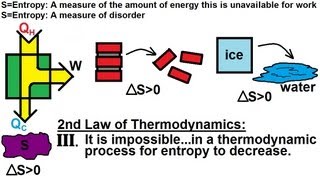
23. The Second Law of Thermodynamics
Entropy and the Second Law of Thermodynamics
Problem 20i
Textbook Question
Textbook QuestionA thin partition divides a container of volume V into two parts. One side contains nA moles of gas A in a fraction fA of the container; that is, VA = fAV. The other side contains nB moles of a different gas B at the same temperature in a fraction fB of the container. The partition is removed, allowing the gases to mix. Find an expression for the change of entropy. This is called the ,entropy of mixing.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
10mPlay a video:
237
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos