20. Heat and Temperature
Specific Heat & Temperature Changes
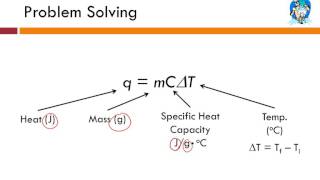
Problem 19.15
Textbook Question
Textbook Question(II) Estimate the Calorie content of 65 g of candy from the following measurements. A 15-g sample of the candy is placed in a small aluminum container of mass 0.325 kg filled with oxygen. This container is placed in 1.75 kg of water in an aluminum calorimeter cup of mass 0.624 kg at an initial temperature of 15.0°C. The oxygen-candy mixture in the small container (a bomb calorimeter, page 545) is ignited, and the final temperature of the whole system is 53.5°C.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
77
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos