23. The Second Law of Thermodynamics
Heat Engines & PV Diagrams
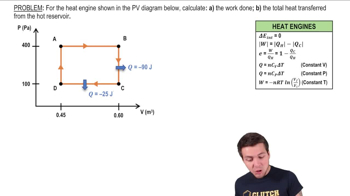
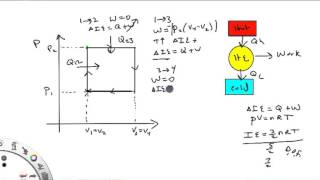
Problem 20.26b
Textbook Question
Textbook Question(II) A heat pump is used to keep a house warm at 22°C. How much work is required of the pump to deliver 2800 J of heat into the house if the outdoor temperature is
(b) - 15°C? Assume a COP of 3.0.
COP = T_H / (T_H - T_L) .
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
45
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos