20. Heat and Temperature
Advanced Calorimetry: Equilibrium Temperature with Phase Changes
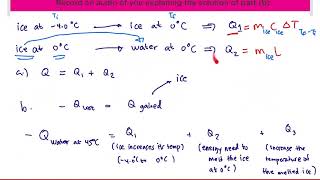
Problem 17.42a
Textbook Question
Textbook QuestionBefore going in for his annual physical, a 70.0-kg man whose body temperature is 37.0°C consumes an entire 0.355-L can of a soft drink (mostly water) at 12.0°C. (a) What will his body temperature be after equilibrium is attained? Ignore any heating by the man’s metabolism. The specific heat of the man’s body is 3480 J/kg K.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
8mPlay a video:
48
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos