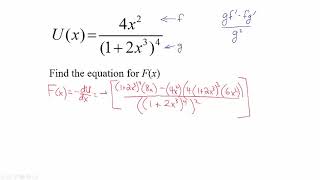10. Conservation of Energy
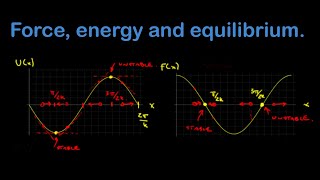
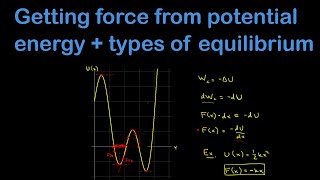
Force & Potential Energy
Problem 8.104a
Textbook Question
Textbook QuestionThe two atoms in a diatomic molecule exert an attractive force on each other at large distances and a repulsive force at short distances. The magnitude of the force between two atoms in a diatomic molecule can be approximated by the Lennard-Jones force, or F(r) = F₀ [2(σ/r)¹³ - (σ/r)⁷], where r is the separation between the two atoms, and σ and F₀ are constants. For an oxygen molecule (which is diatomic) F₀ = 9.60 x 10⁻¹¹ N and σ = 3.50 x 100⁻¹¹ m .
(a) Integrate the equation for F(r) to determine the potential energy U(r) of the oxygen molecule.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
155
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos